It is antiferromagnetic with a Néel temperature of 475 K. However, ferrochrome as a starting material requires a prior separation of the iron.Įxtremely pure chromium is produced by further purification steps using the van-Arkel-de-Boer process.įerrochrome is produced by reducing chromite in an electric arc furnace at 2800 ° C.Ĭhromium is a silver-white, corrosion- and tarnish-resistant hard metal that is tough, malleable and malleable in its original state. Corresponding solutions are prepared by dissolving chromium (III) oxide or ferrochrome in sulfuric acid. Purer chromium is made by electrodepositioning the Cr 3+Ions from sulfuric acid solution shown. This is followed by the aluminothermic reduction of the chromium (III) oxide to chromium:Ĭhromium cannot be obtained from oxidic ores by reduction with coal, since this creates chromium carbide. A subsequent reduction with carbon gives chromium (III) oxide: The sodium dichromate crystallizes on cooling as a dihydrate from the solution. The sodium chromate is extracted with hot water and converted into dichromate with sulfuric acid: In the second step, an oxidizing digestion takes place at approx. The extracted chromite ore is freed from the dead rock. The compounds of chromium have many different colors and are often used as pigments in paints and varnishes. It belongs to the transition metals, in the periodic table it is in the 6. Χρῶμα chrṓma, color ') is a chemical element with the element symbol Cr and the atomic number 24. 
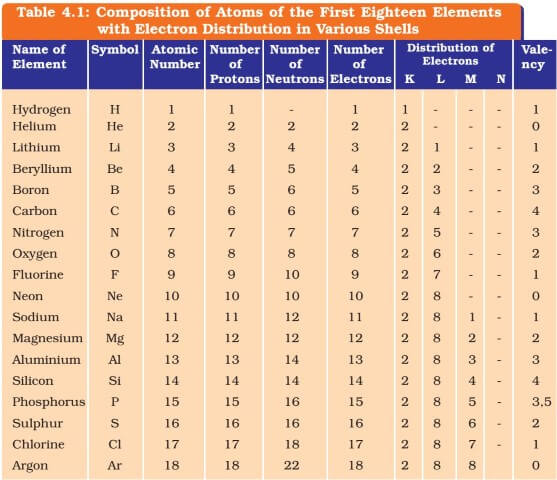
Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.Chromium (alt. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
